Wikipedia is my source of choice for up-to-date summaries of biomedical information. I queried “leukotrienes” and immediately ran across the original name for these inflammatory compounds, “slow reacting substance of anaphylaxis”. I was initially distracted by the classic experimental use of snake venom and histamine to induce leukotriene production. Snake venom has the same enzyme, PLA2, as brown recluse spider venom (subject of a previous article) and honey bee venom, that releases arachidonic acid (ADA). ADA is an omega-6 fatty acid that is the starting material for inflammatory prostaglandins and leukotrienes.
 The mention of honey bee venom in the Wikipedia article on anaphylaxis sent me on a quick check of the structure and sequence of the honey bee allergen. I initially found that the major allergen is a hyaluronidase. I quickly searched for a three amino acid sequence that I predicted would make it an allergen.
The mention of honey bee venom in the Wikipedia article on anaphylaxis sent me on a quick check of the structure and sequence of the honey bee allergen. I initially found that the major allergen is a hyaluronidase. I quickly searched for a three amino acid sequence that I predicted would make it an allergen.  It was just where I expected to find it. About two thirds of the way along the amino acid sequence I found, -TTSRKKVLP-. Three basic amino acids together, in this case -RKK-, argininine-lysine-lysine, form a strong heparin-binding domain, that I believe takes proteins into cells and during inflammation primes the immune system for allergic responses.
It was just where I expected to find it. About two thirds of the way along the amino acid sequence I found, -TTSRKKVLP-. Three basic amino acids together, in this case -RKK-, argininine-lysine-lysine, form a strong heparin-binding domain, that I believe takes proteins into cells and during inflammation primes the immune system for allergic responses.I have found the same strong heparin-binding domain associated with allergens of ragweed, dust mites and peanuts. The principal autoantigens of autoimmune diseases, such as lupus, celiac, etc. also display the same unusual sequences. In lupus, or example, nuclear proteins with the internalization signal provided by nucleic acid-binding domains (and nuclear localization signas) are autoantigens. This pattern is found with all allergens that I have examined. There are a few apparent exceptions, but in all of these cases, there is a closely related allergen from a related source that has the expected strong heparin-binding domain. It appears that in these cases, the less common allergen provides the initial exposure during the presentation phase of high inflammation, and the allergy is maintained by subsequent exposure to the more common allergen. After the establishment of the allergy, the strong heparin-binding domain is no longer needed, because antibodies bind to other parts of homologous allergenic proteins for internalization.
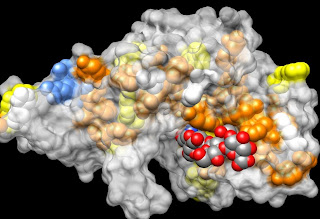
Just for fun, I have illustrated the honey bee allergen, hyaluronidase, to show both its strong heparin-binding domain (blue) along with its substrate hyaluronan (grey and red). Note that the substrate sugars are in the slot of the active site, which is lined with orange and yellow aromatic amino acids that provide flat, hydrophobic binding platforms for each sugar.
After this little distraction to provide further support for my explanation of the cause of allergies, I have to get back to looking at the role of leukotrienes in anaphylaxis, COPD, asthma and other inflammatory diseases.

3 comments:
I took a stab that there was likely an older article on here, dissecting this very same premise and -- voila!
is there a prevention for bee stings to avoid allergic reactions? my daughter is highly allergic to pollen as well as a long list of fruit and vegetables (IgE not intolerances) would probiotics help in this case too? Claudia
I enjoyed this post thanks for sharing
Post a Comment